SciMed Education
Liposome synthesis & size characterisation using microfluidics & high‑precision DLS
In Summary
Liposomes are spherical nanoparticles made from lipid bilayers that can encapsulate both water‑soluble and lipid‑soluble compounds, making them essential vehicles for drug delivery, cosmetics and diagnostics.
Traditional batch synthesis methods often produce liposomes with inconsistent sizes and low encapsulation efficiency, but modern microfluidic platforms such as the TAMARA system, when combined with high‑precision dynamic light scattering (DLS) measurement, enable highly reproducible production of monodisperse liposomes with finely controlled size and polydispersity.
What are liposomes and why are they important in drug delivery and diagnostics?
Liposomes are hollow, spherical vesicles formed from one or more concentric phospholipid bilayers, resembling tiny bubbles. Because the lipid bilayer encloses an aqueous compartment, liposomes can encapsulate hydrophilic compounds inside the vesicle and lipophilic compounds within the membrane itself. This versatility has made them invaluable across medicine, cosmetics and diagnostics: they can tailor payload release rates and improve bioavailability, enabling controlled delivery of therapeutic molecules and contrast agents.
The ability to adjust their composition allows researchers to customise stability, release kinetics and targeting, positioning liposomes as a key component of modern pharmaceutical research and clinical practice.
Why do conventional liposome synthesis methods need improvement?
Traditional liposome preparation techniques typically use batch processes such as film hydration and homogenisation.
These methods often suffer from limited control over particle size distribution and encapsulation efficiency, leading to significant batch‑to‑batch variability and even structural damage to the vesicles. In conventional homogenisation, high shear forces may destabilise the liposomes, and the poor control over mixing creates broad particle size distributions that compromise drug loading and release characteristics.
Because liposome size is a critical quality attribute affecting stability and cellular uptake, the limitations of batch methods underscore the need for innovative production technologies that offer precise control and reproducibility.
How does microfluidic mixing enable precise liposome synthesis?
Microfluidic platforms have emerged as powerful alternatives to batch manufacturing because they control fluid dynamics within microscopic channels. In microfluidic devices, two miscible streams – typically a lipid solution in an organic solvent and an aqueous buffer – meet and mix rapidly.
The sudden change in solvent environment reduces lipid solubility and triggers self‑assembly into nanoparticles. The mixing speed directly influences particle size: faster and more homogeneous mixing produces smaller, more uniform liposomes, whereas slower mixing yields larger and broader distributions.
Microfluidic channels maintain laminar flow and constant mixing times, delivering excellent reproducibility and allowing users to tune flow rates and ratios to achieve target sizes and narrow polydispersity. The TAMARA platform exemplifies this approach; by carefully controlling flow rates and chip geometry it allows researchers to adjust nanoparticle size with unparalleled precision.
Why is nanoparticle size and polydispersity index critical in lipid nanoparticle formulations?
The size of lipid nanoparticles determines many of their biological properties. Smaller particles generally have longer circulation times and improved cellular uptake, but particles that are too small may exhibit toxicity, while those larger than about 200 nm risk rapid clearance by the liver or filtration by the kidneys.
Most lipid nanoparticles used for therapeutic delivery fall between 60 nm and 120 nm, balancing stability, payload capacity and biodistribution. Not only the average size but also the width of the size distribution matters. The polydispersity index (PDI) quantifies the degree of non‑uniformity within a nanoparticle population; it is calculated from the ratio of the standard deviation to the mean size and squared.
A lower PDI indicates a more monodisperse sample, which translates to predictable pharmacokinetics and reduced toxicity risks. Regulatory guidelines recommend keeping the PDI below 0.3 for liposomal drug products, and advanced microfluidic techniques like TAMARA regularly achieve PDIs around 0.1 for siRNA‑loaded lipid nanoparticles.
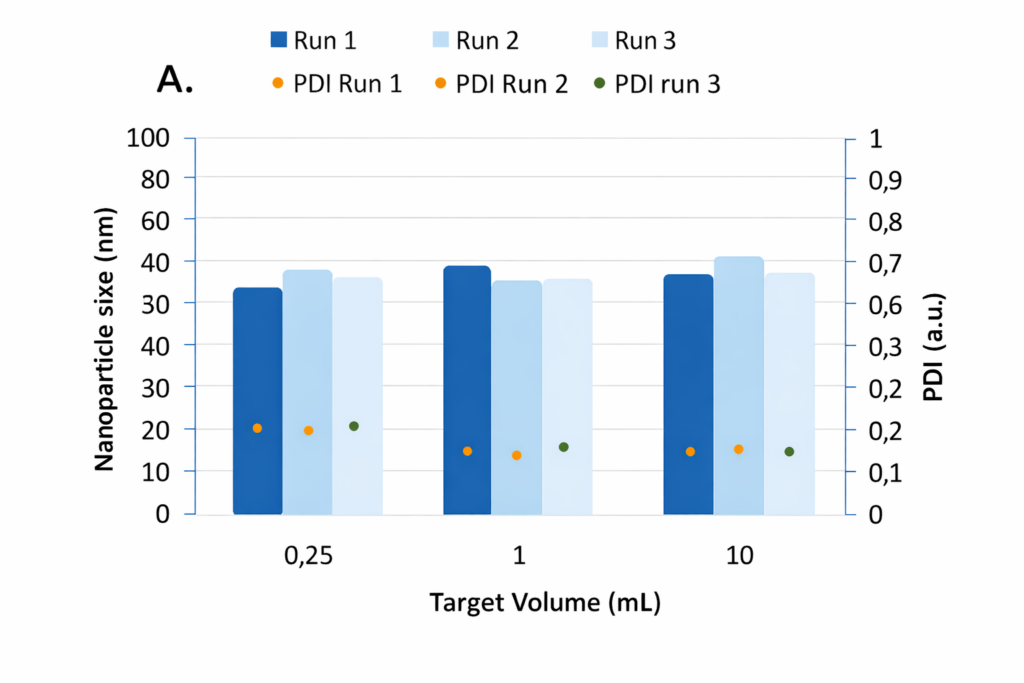
- Nanoparticle size and polydispersity index (PDI) measured across three runs at different target volumes, demonstrating consistent particle size and low PDI during liposome synthesis.
What is dynamic light scattering and how does it characterise liposomes?
Dynamic light scattering (DLS) is a widely used technique for measuring the size of particles suspended in a fluid. When a laser beam passes through a suspension, particles undergoing Brownian motion scatter the light.
Fluctuations in the scattered light intensity are directly related to the diffusion coefficient of the particles, which can be converted into a hydrodynamic radius using the Stokes–Einstein equation. DLS is rapid, non‑invasive and capable of analysing a broad range of particle sizes, making it particularly suitable for liposome suspensions.
Accurate results depend on appropriate sample concentration and dilution; studies have shown that liposome concentration during DLS measurements significantly affects the reliability of the size determination.
How were liposomes synthesised and characterised using the TAMARA and VASCO Kin platforms?
In the experiments described, the lipids used were phosphatidylcholine (Lipoid S100) and cholesterol at a mass ratio of 2:1. A stock solution of 60 mg mL⁻¹ was prepared by dissolving the lipid powders in absolute ethanol with sonication. This stock was diluted to a working solution of 10 mg mL⁻¹, and demineralised water served as the aqueous phase during synthesis. Using the TAMARA platform, researchers selected the synthesis conditions on the controller, pipetted the reagents into reusable reservoirs and initiated the microfluidic mixing. After a few seconds the liposome suspension was collected and stored at 4 °C until measurement.
Prior to DLS analysis, samples were diluted twenty‑fold in deionised water. Size characterisation employed the VASCO Kin DLS instrument, which features a sensitive single‑photon avalanche photodiode detector and fast acquisition electronics for real‑time monitoring. The instrument’s patented correlator enables nanometre‑scale measurements under static and dynamic conditions while suppressing artefacts such as dust. For these experiments, an in‑situ measurement head connected via optical fibres allowed direct, contactless analysis without the need for sample batching.
What do batch‑to‑batch repeatability tests tell us about the TAMARA platform?
To assess reproducibility, liposomes were synthesised under constant conditions using a herringbone microfluidic mixer. The flow‑rate ratio between aqueous and organic phases was fixed at 3:1 with a total flow rate of 4 mL min⁻¹, and the final target volumes were varied (0.25 mL, 1 mL and 10 mL), each in triplicate. After each run the chip was carefully cleaned.
Results showed excellent repeatability across all volumes: the relative standard deviation in size was 6.5 % at 0.25 mL, 5.5 % at 1 mL and 2.5 % at 10 mL, corresponding to a maximum size difference of about 4 nm. These variations fall well below the typical ±5 % repeatability of DLS measurements, indicating that the microfluidic synthesis itself is highly reproducible. All samples were homogeneous, with PDIs below 0.3 at the smallest volume and below 0.2 at larger volumes. Slightly higher PDI at very small volumes was attributed to the greater proportion of the initial and final fractions of the run where mixing is less optimal.
"Advancing Liposome and Lipid Nanoparticle Characterisation
with High Precision DLS"
How does real time monitoring enhance DLS measurements of liposomes in the field of drug delivery and diagnostics?
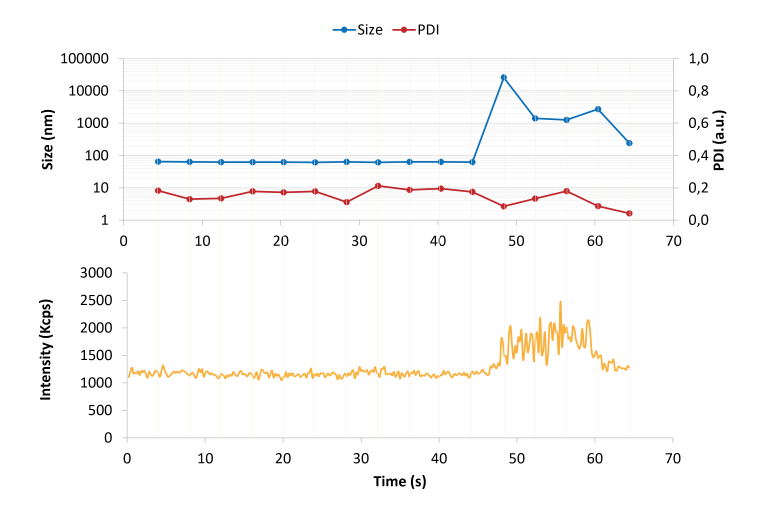
The VASCO Kin instrument includes NanoKin software that enables real‑time tracking of particle size during measurements. In the example provided, an unexpected event occurred during a one‑minute measurement.
By observing the intensity trace, researchers identified the anomaly as a dust particle or fibre typical of non‑cleanroom environments. Such contaminants, often micrometre‑sized, can distort DLS results. The software can automatically detect these artefacts and remove their contributions, ensuring that the calculated size distribution reflects only the sample. This capability allows continuous monitoring of liposome synthesis processes and provides confidence that occasional dust events do not compromise data quality.
How do TAMARA and VASCO Kin advance liposome synthesis and drug delivery?
The combined microfluidic and DLS approach offers considerable advantages over traditional methods. The TAMARA platform delivers precise control over liposome size and monodispersity, addressing key limitations of conventional batch processes. Its ability to consistently produce liposomes with optimal PDIs enhances the efficacy and safety of drug delivery systems.
Coupled with the VASCO Kin instrument, which provides real‑time monitoring and reproducible size measurements, the system ensures uniform, high‑quality liposome samples suitable for pharmaceutical and medical applications. Together, these technologies represent a significant advance in liposome synthesis and characterisation, paving the way for more reliable and effective nanomedicine products.
What to do Next?
If you are working with liposomes, lipid nanoparticles, or other nanomedicine formulations, precise particle size measurement is essential for ensuring formulation stability, reproducibility, and drug delivery performance. Explore our particle size characterisation solutions or speak with one of our expert product managers to discuss the best dynamic light scattering systems for your application.
Page FAQ's
A liposome is a spherical vesicle composed of one or more lipid bilayers. Its structure allows both hydrophilic and lipophilic molecules to be encapsulated, making liposomes useful carriers for drugs, cosmetics and diagnostic agents.
Microfluidic mixing controls the flow of liquids in microscopic channels, ensuring rapid and homogeneous mixing. This approach produces liposomes with narrow size distributions and high reproducibility, overcoming the variability and low encapsulation efficiency of conventional batch methods.
PDI quantifies the breadth of particle size distribution within a sample. A lower PDI indicates a more uniform (monodisperse) population, which is desirable for predictable drug delivery. Regulatory guidance recommends keeping PDI below 0.3, and microfluidic platforms can routinely achieve much lower values.
DLS measures fluctuations in the intensity of light scattered by particles undergoing Brownian motion. These fluctuations reveal the diffusion coefficient, from which the hydrodynamic diameter of liposomes is calculated. DLS offers a rapid, non‑invasive means of sizing nanoparticles across a broad range.
TAMARA’s microfluidic design allows precise control over liposome size and polydispersity, while the VASCO Kin DLS instrument provides sensitive, real‑time size measurements with artefact rejection. Together they enable reproducible synthesis and characterisation of liposomes suited to pharmaceutical and research applications.
Contact Us Today
We take great pleasure in assisting you and ensuring you get a prompt response to your questions
Live chat opening hours Mon – Fri 9:15 to 16:30 (UK Time)